Background
Huntington’s Disease causes progressive motor dysfunction through atrophic disruption to the frontal cortical motor circuitry and basal ganglia regions. Symptom onset typically begins at age 40; subsequent neurodegeneration is incurable and terminal. Motor Response Potential (MRP) studies examining symptomatic patients have identified reduced peak neural amplitude and pre- and post-slope components compared with controls during simple movements such as finger tapping. However, little is known about the profile of MRPs in presymptomatic patients, who show no significant behavioural differences in response time from healthy controls. Atypical MRPs in presymptomatic patients may be a potential biomarker for the disease that could be useful for pharmaceutical trials.
Methods
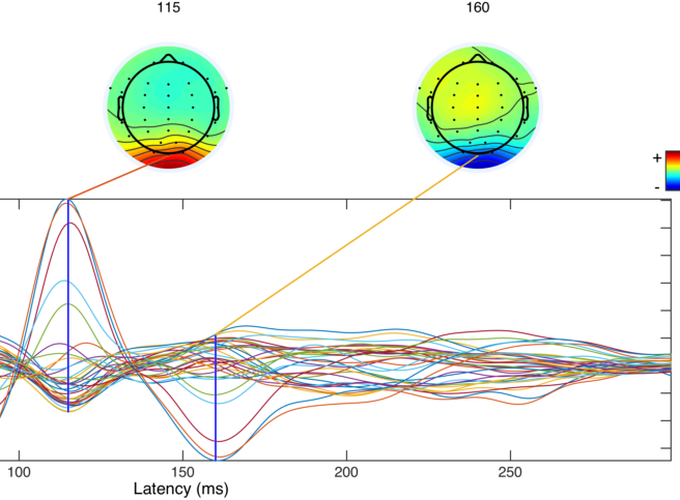
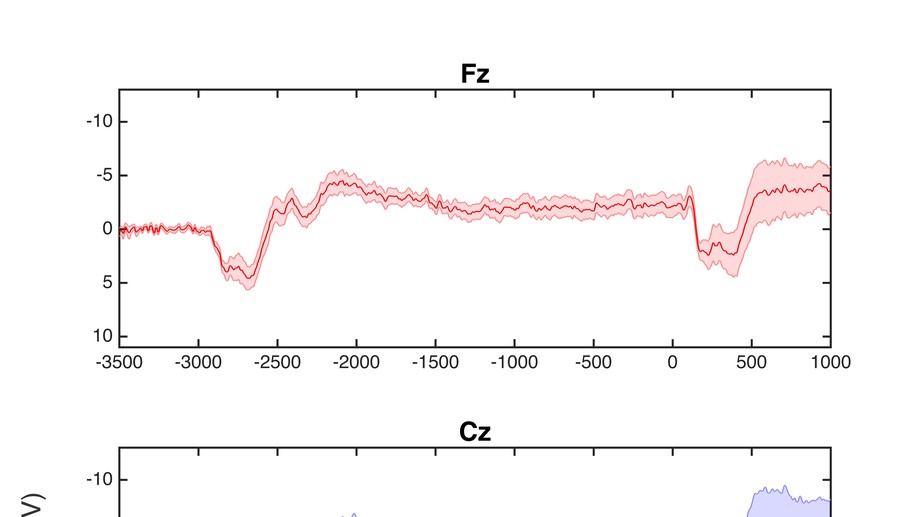
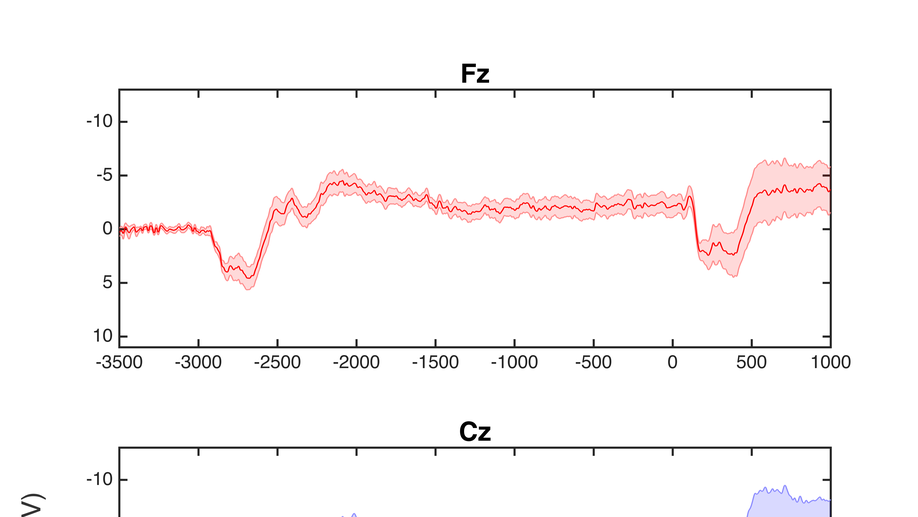
10 genetically confirmed symptomatic HD participants, 17 genetically confirmed presymptomatic participants (PHD), and 17 healthy controls were recruited for this study. Participants completed a sequential tapping task using their right index finger, alternating key taps (left and right) at a rate of one every four seconds. In a cued tapping task, visual cues indicated the appropriate response times, and in self-paced tapping, the participant continued alternating taps at the same rate in the absence of cues. Conditions alternated in 24-second segments, and continued for a total task period of 8 minutes. Experimental variables included Contingent Negative Variation (CNV) magnitude (peak and area under the curve), slope and intercept, and amplitude (peak to peak) and latency of the motor response (-50 to +100 ms, and 100 to 300 ms relative to the response).
Results
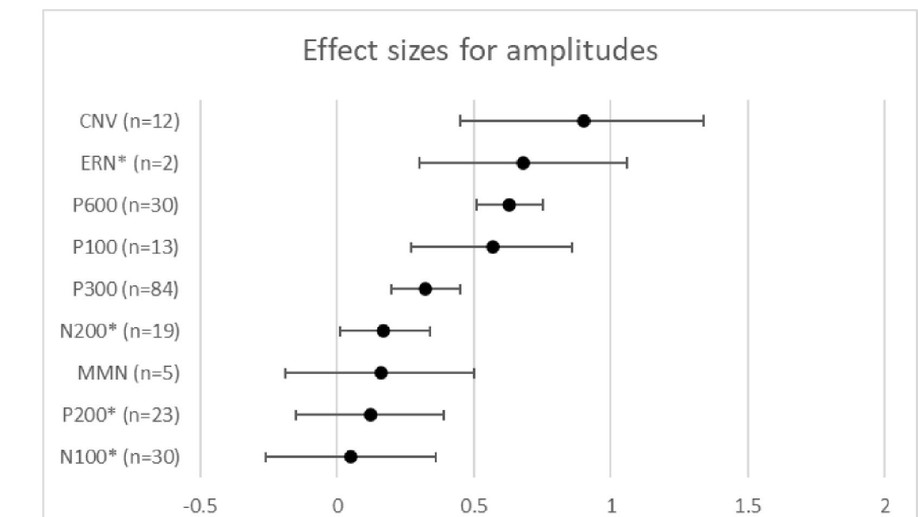
There was no amplitude difference between groups for peak or area under the curve CNV, or CNV slope or intercept. A significant group difference was found in the motor response peak to peak amplitude (F = 8.844, p = .001) and latency (F = 4.195, p = .022). Post hoc analyses revealed significantly larger amplitudes for controls than HD (p < .001), and PHD than HD (p =.041), and significantly later latencies for controls than HD (p = .020).
Discussion
Presymptomatic patients show no significant difference in behavioural performance tasks using measures such as reaction time. This study found that MRP s of PHD participants varied markedly in profile, but were not significantly different from controls. Observed variability may indicate functional differences in the neural generation of motor responses during early stages of the disease. Additional analysis will be conducted to explore the relationship between PHD MRP profiles and other variables such as predicted time to onset, age, and number of CAG repeats.